|
12/18/2022 0 Comments Density altitude 
The International Standard Atmosphere (ISA) model assumes a lapse rate of 1.98☌ per thousand feet. So, the small term aLH 0 is subtracted from 1, then multiplying that number times the exponential solution LH 0 gives the corrected solution. 1) h/145,454 = 1 - T/T 0 = 1 - (T 0/T) -1Īn approximate solution to the barometric equation using the temperature formula in H is h ≈ LH 0 (1 - aLH 0), where 1/a = 290,909. Temperature is T 0 = 15☌ = 288° Kelvin, pressure is p 0 = 29.92" (inches of Hg), and the temperature drops 1.98☌ (or °K) each 1000' this is the lapse rate. So the International Standard Atmosphere (ISA) model is used. If the pressure were half of sea level pressure, then h = 27,500 ln 2 = 19,062'.Ī solution to the equation when H is not constant, but changes with temperature, requires an equation for temperature as a function of height. Inverting and taking the natural logarithm of both sides gives h = H 0L where L is the natural log of the pressure ratio L = ln (p 0/p). The pressure is dropping exponentially with height. Sea level pressure is p 0 = 29.92" and there, H = H 0. If H were constant there is a simple exponential solution found by integrating this equation the pressure at the aircraft at height h is p = p 0 exp. This is a scale height factor for altitude. Note that H is proportional to T, which changes (decreases) with altitude in the Troposphere, and at sea level H = H 0 = 27,500'. Finally, T is the temperature in °K (degrees Kelvin = degrees centigrade + 273☌). That mass is about 29 times the mass of a proton, determined by the mix of oxygen and nitrogen and The acceleration due to gravity, k is the Boltzmann constant, and m is the average mass of the air molecules in aĬolumn of air. The result is the differential form of the barometric equation, H dp = - p dh, where H is a collection of parameters defined by mgH = kT. 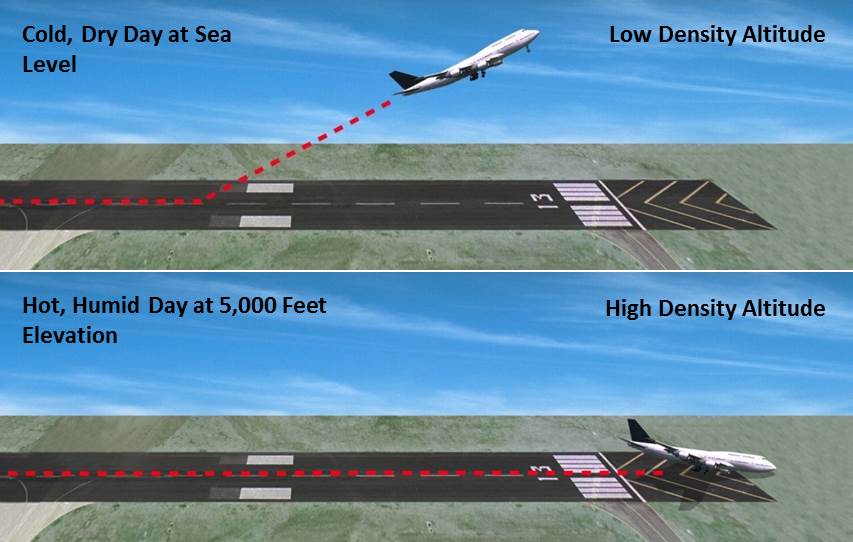
From that force balance a simple relationship can be written for the very small change in pressure (dp) at your location caused by that very smallĬhange in elevation (dh). The gravity force of the air in the slice is balanced by the reduction in pressure between the top and bottom of the slice. The Barometric Law is derived by equating forces on a very small slice of air with thickness dh at altitude h. International standard used in aviation, so it is important to understand how your altimeter does this, and to know why there are errors in this process and how big they might be. Even though we can nowĭetermine our altitude far more accurately from a WAAS GPS position solution, barometric altitude is still the With that model you can determine your altitude from the static pressure at the aircraft. This profile cannot be known, so we represent the atmosphere by the so-called International Standard Atmosphere (ISA) model. Toįind a solution you need to know the temperature profile from the ground to the aircraft. It relies on the Barometric Law, an equation that can be solved for pressure changes with height above the ground. In any case, the declared data was a bit rough and ready, anyway.įor the exams, I'm sure that the examiner will figure out something to suit what he did previously with the declared charts.In aviation, altitude is estimated from the static pressure measured by your altimeter. Keep in mind that the declared data was developed in ancient times for a need when communications were problematic for much of the time. With communications capabilities these days, the need for something like the declared data has been pretty much sidelined. 
Doesn't seem to contain the declared density altitude chartsĪppears they have been consigned to the waste bins of history.įrom a practical point of view, you can get adequately accurate elevations from the net - eg, see the other relevant thread atī/forum/performance/6951-declared-density-chartsīetween the ASA and BOM sites you can get MSL pressures and reasonably accurate OAT forecasts.Ĭombine these and you have a reasonable solution for light aircraft planning in the event that you can't contact someone to obtain actual temperatures and QNH. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
